
The U.S Food and Drug Administration (FDA) has approved a human trial of a new treatment for HIV.
Currently, antiretroviral drugs are the most effective treatment for the virus. If HIV positive people take the medication, they can expect to live a normal lifespan.
However, although the drugs are effective in reducing viral load, if you stop taking them, the virus can replicate and wreak damage. Individuals can also sometimes develop resistance to the medication or experience side effects.
Related: Scientists can’t find any trace of HIV in man potentially “cured” via medication alone
How about we take this to the next level?
Our newsletter is like a refreshing cocktail (or mocktail) of LGBTQ+ entertainment and pop culture, served up with a side of eye-candy.
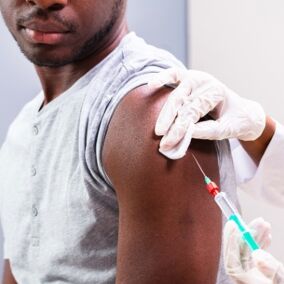
The new trial will test a gene therapy developed by biotech firm American Gene Technologies (AGT), based in Rockville, Maryland.
Its HIV gene therapy is known as AGT103-T. It involves removing some white blood cells from an HIV-positive patient, then inserting a gene that modifies the T-cells. This enables the cells to stop HIV replication.
The HIV-resistant CD4 T-cells are then infused back into the patient, where they can duplicate and fight HIV. A more-detailed, peer-reviewed paper on AGT103-T appeared in June in the journal, Molecular Therapy.
Jeff Galvin, founder and chief executive officer of AGT, said in a statement, “I am confident AGT103-T will be an important step towards an eventual cure for HIV.”
The company’s chief science officer, David Pauza, added, “This is momentous news that we have FDA approval to launch Phase I and conduct our first human trials. We are beyond excited to reach this milestone. This brings us closer to our goal of transforming lives with genetic medicines. Based on our successful commercial-scale product manufacturing runs and features of the product observed in our labs, this therapy has a high potential to be effective.”
If successful, the company hopes the gene therapy will reduce or even eliminate the need for HIV-positive patients to take further HIV medication.
The Phase 1 trial will be conducted in the Baltimore and Washington DC area, specifically at Washington Health Institute, University of Maryland, Institute of Human Virology and Georgetown University.
Speaking to Queerty, Galvin said the phase 1 trial will treat six patients and researchers will look for specific biological signs the treatment is working. If all goes well, it might be extended to further patients and the next step would be to then seek approval from the FDA and the test subjects to suspend their antiretroviral therapy, to see if the gene therapy has prompted long term remission or if they have been “functionally cured,” according to Galvin. If so, in terms of health significance, “that would be huge,” he said.
He said he hopes a phase 2 trial, recruiting further patients, could be launched before the end of next year if the treatment works and is shown to be safe.
Related: Major HIV vaccine trial abandoned because the vaccine doesn’t work
“The big question everyone has is, ‘when will it be available for me?’,” said Galvin. “I think it’s all dependent on demonstrating human efficacy, but we’ve already talked to five different pharma companies that are interested in this project. There is some history of gene therapy being licensed out of a pivotal phase 2 for acute conditions, and I think the FDA is quite concerned about people that can’t tolerate antiretroviral therapy. I wouldn’t be surprised, if phase 2 is successful, there might be some limited license that might come out of that.
“If not, I would expect that it would be another two years past the end of phase 2, so we’re talking 3-4 years from now as a minimum.”
Those wishing to be considered for the trial must have taken antiretroviral therapy (ART) for 1-3 years. They must have undetectable virus levels and a stable CD4 T-cell count above 500 cells/mm3, and may not have AIDS-defining conditions.
Enrolment will begin in September and AGT hopes to have some initial results by the end of the year.









































Vince
Sounds very promising. Even a partial success would be a game changer.
Seems like every 5 years major changes are happening. First the one pill a day. Now the once a month Injectables.
iron
regardless of what the meds do. remember treatment is not a cure l think.in terms of vaccine if it works for a while it is a treatment. not a cure enough said
evanxx
Only HIV/poz people are included in the trials? Why are long-term AIDS survivors excluded. Like all recent changes, the ones who waited the longest and need it most; “it’s too little & too late”. We were kicked to the curb a long time ago. People with AIDS (PWA’s) might as well have died in the 80’s. U=U and PrEP are also examples of research that only benefits younger people.
#endAIDSstigma #WeWontBeErased
Chrisk
I would imagine it requires coming off medication. Very problematic for someone who has aids. They want healthy bodies which makes sense for liability reasons.
U=U and PrEP isn’t limited to just young people.
pharaon.em.joe
Bunch of nonsense.
Den
1. PrEP is specifically for HIV negative people, to prevent spread of HIV, especially in couples where one is positive and one is negative. Why exactly are you bitter about that? Seems odds to me.
2. And what exactly bothers you about the notion that undetectable viral loads prevent people from infecting others? What is the source of your belief that long term survivors cannot achieve an undetectable viral load? They can, and do, with antiretroviral drugs.
3. As for the specifics of the cohort for the small phase one trial. ALL criteria must be uniformly met by all participants for results to have statistical significance. People with opportunistic infections or fluctuating voral loads will be taking other drugs, and that will skew results.
Clinical trials MUST be carefully and precisely controlled or they are pointless.
Chrisk
@Den
Evan has always been on the bitter side of things in these kind of posts. I understand where it’s coming from so I usually give him some respect. Plus, nothing wrong with a little pessimism.
Seaman 51
No disrespect to Wilson Cruz, but don’t you think this might have been a more significant leading story?
Den
This is Queerty. Flesh always leads.
Cam
Let me guess, in Europe, Canada, Coasta Rica, etc. the treatment will cost ten dollars per month. In America it will be ten thousand dollars.
Chrisk
Big Pharma is going to get a hold of this and twist the patent to make sure no one else can do it without paying big $. That’s how it usually starts.
frankcar1965
Big pharma is in bed with your politicians which the fascist Republicans put in office, you must vote them out to get results, now that the Democrats are in control, maybe voting reform can get the money out of our elections.